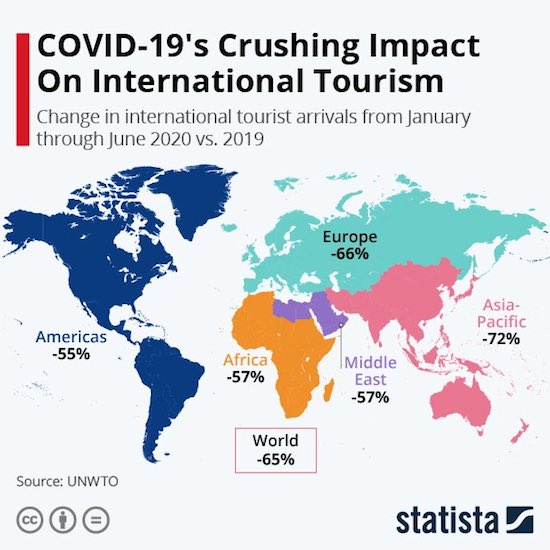
Edvard Munch Spring in Johan Karl Street 1944

Another one of those days where there’s just too much news. So I split it up in this Covid Rattle and a Debt Rattle that will follow imminently.

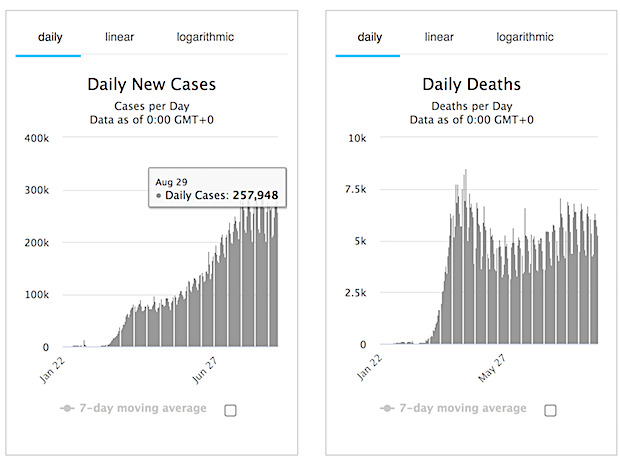
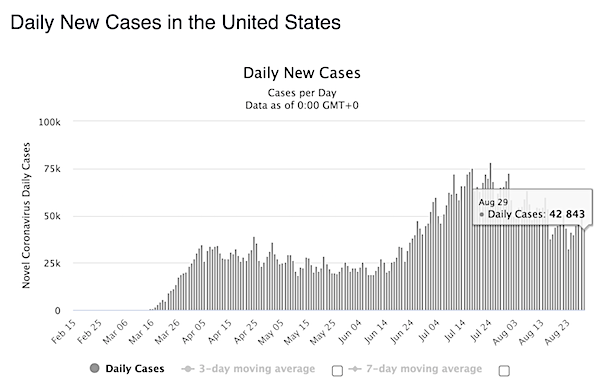
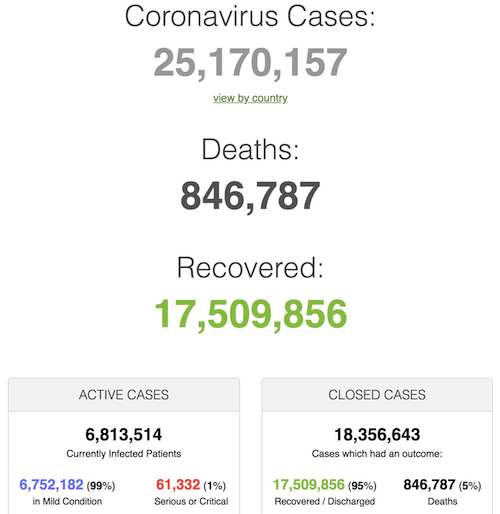
Despite all the bad news, here’s what I think will happen in much of the northern hemipshere, following this graph from Daniel J. Wilson at the Federal Reserve Bank of San Francisco (see Debt Rattle April 13)
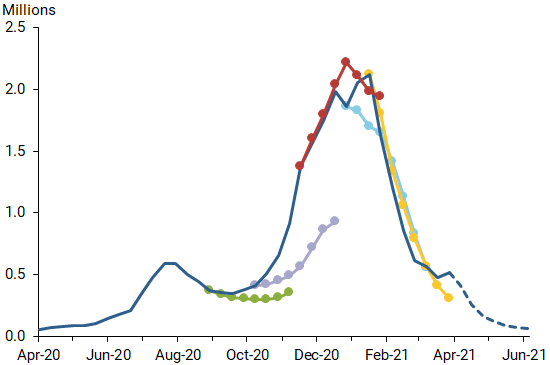
Around June 1, in many countries the viral load will have fallen off a cliff. Summer will be much less dangerous than winter. Makes sense. Whether Covid will return in autumn will depend largely on what we do to make people’s immune systems stronger. Vitamin D plays a large role in that, but so do exercise and healthy food.

Just as everyone starts banning AstraZeneca, turns out mRNA “vaccines” are just as bad. And just as poorly tested.
• Blood Clots As Prevalent With Pfizer, Moderna Vaccine As Astrazeneca (MW)
A study by Oxford University found the number of people who receive blood clots after getting vaccinated with a coronavirus vaccine are about the same for those who get Pfizer and Moderna vaccines as they are for the AstraZeneca vaccine that was produced with the university’s help. According to the study, 4 in 1 million people experience cerebral venous thrombosis after getting the Pfizer or Moderna vaccine, versus 5 in 1 million people for the AstraZeneca vaccine. The risk of getting CVT is much higher for those who get COVID-19 — 39 in a million patients — than it is for those for get vaccinated. AstraZeneca’s vaccine use has been halted or limited in many countries on blood clot concerns.

But the report this is based on comes from Oxford, one of the makers of the AstraZeneca vaccine.
• Risk Of Rare Blood Clotting Higher For Covid-19 Than For Vaccines (Oxford)
Researchers at the University of Oxford have today reported that the risk of the rare blood clotting known as cerebral venous thrombosis (CVT) following COVID-19 infection is around 100 times greater than normal, several times higher than it is post-vaccination or following influenza. The study authors, led by Professor Paul Harrison and Dr Maxime Taquet from Oxford University’s Department of Psychiatry and the NIHR Oxford Health Biomedical Research Centre, counted the number of CVT cases diagnosed in the two weeks following diagnosis of COVID-19, or after the first dose of a vaccine. The then compared these to calculated incidences of CVT following influenza, and the background level in the general population.
They report that CVT is more common after COVID-19 than in any of the comparison groups, with 30% of these cases occurring in the under 30s. Compared to the current COVID-19 vaccines, this risk is between 8-10 times higher, and compared to the baseline, approximately 100 times higher. The breakdown comparison for reported cases of CVT in COVID-19 patients in comparison to CVT cases in those who received a COVID-19 vaccine is:
• In this study of over 500,000 COVID-19 patients, CVT occurred in 39 in a million patients.
• In over 480,000 people receiving a COVID-19 mRNA vaccine (Pfizer or Moderna), CVT occurred in 4 in a million.
• CVT has been reported to occur in about 5 in a million people after first dose of the AZ-Oxford COVID-19 vaccine.
• Compared to the mRNA vaccines, the risk of a CVT from COVID-19 is about 10 times greater.
• Compared to the AZ-Oxford vaccine, the risk of a CVT from COVID-19 is about 8 times greater.However, all comparisons must be interpreted cautiously since data are still accruing. Paul Harrison, Professor of Psychiatry and Head of the Translational Neurobiology Group at the University of Oxford, said: ‘There are concerns about possible associations between vaccines, and CVT, causing governments and regulators to restrict the use of certain vaccines. Yet, one key question remained unknown: ‘What is the risk of CVT following a diagnosis of COVID-19?’. ‘We’ve reached two important conclusions. Firstly, COVID-19 markedly increases the risk of CVT, adding to the list of blood clotting problems this infection causes. Secondly, the COVID-19 risk is higher than we see with the current vaccines, even for those under 30; something that should be taken into account when considering the balances between risks and benefits for vaccination.’

Pfizer is winning.
• EU Commission To End AstraZeneca and J&J Vaccine Contracts At Expiry (R.)
The EU Commission has decided not to renew COVID-19 vaccine contracts next year with AstraZeneca and Johnson & Johnson (J&J), Italian daily La Stampa reported on Wednesday, citing a source from the Italian health ministry. “The European Commission, in agreement with the leaders of many (EU) countries, has decided that the contracts with the companies that produce (viral vector) vaccines that are valid for the current year will not be renewed at their expiry,” the newspaper reported. It added that Brussels would rather focus on COVID-19 vaccines using messenger RNA (mRNA) technology, such as Pfizer’s and Moderna’s.
A spokesman for the EU Commission said it was keeping all options open to be prepared for the next stages of the pandemic, for 2022 and beyond. “We cannot, however, comment on contractual issues,” the spokesman added. Later on Wednesday the President of the European Commission said the EU was in talks with Pfizer and BionTech for a new contract for 1.8 billion doses, confirming a Reuters report from last week. “We need to focus on technologies that have proven their worth. mRNA vaccines are a clear case in point,” she added.

India is not winning. Wonder what went wrong there. Note: 1.4 billion is a lot of people.
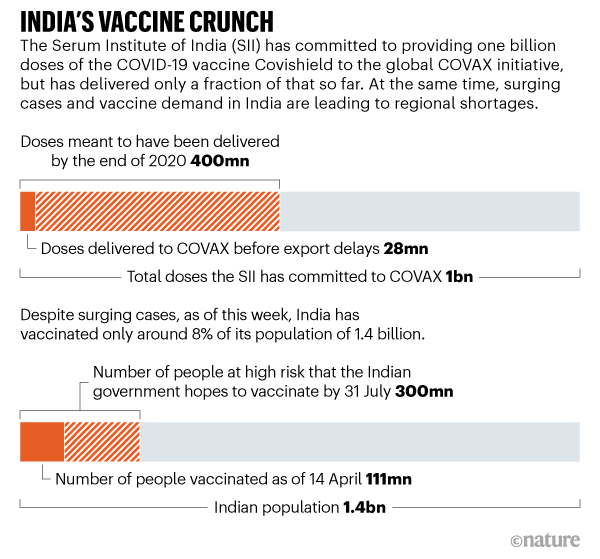
• India’s COVID-Vaccine Woes — By The Numbers (Nature)
India, one of the world’s biggest suppliers of vaccines, is facing a COVID-19 vaccine crunch, partly due to an explosion of cases linked to new variants. This spells trouble for many countries relying on Indian-made vaccines supplied through the World Health Organization’s COVAX initiative for equitable access to vaccines. On 12 April, India reported 168,912 new COVID-19 cases, its highest daily total yet. It has now had more than 13.5 million confirmed cases in total, overtaking Brazil as the world’s second-worst hit country, behind the United States. “While new variants are surfacing in India, a decline in COVID-appropriate behaviour such as wearing masks and social distancing is adding to their faster spread,” says Randeep Guleria, director of the All India Institute of Medical Sciences in New Delhi.
By 14 April, more than 111 million people had been vaccinated in the country (see ‘The race to vaccinate’). But in March, fears of vaccine shortages led to the government temporarily halting exports of a version of the University of Oxford–AstraZeneca vaccine known as Covishield, which is produced by the Serum Institute of India (SII), based in Pune. The SII, the world’s largest manufacturer of vaccine components, was expected to provide many of the doses for COVAX. But a fire at a facility in January is part of the reason that it hasn’t yet been able to live up to its production target of 100 million doses per month. Currently it produces between 60 million and 65 million per month.
Last June, AstraZeneca, which is based in Cambridge, UK, announced that it had licensed the SII to supply a total of one billion doses of Covishield for low- and middle-income countries. But only 64 million had been exported before the halt in exports last month, 28 million of which went to COVAX. Earlier this year, chief executive Adar Poonawala said on Twitter that the SII had been directed by the Indian government “to prioritise the huge needs of India and along with that balance the needs of the rest of the world”. India’s battle with a surge in cases could delay planned deliveries of Covishield to 64 lower-income countries through COVAX, according to a 25 March statement by initiative member Gavi, the Vaccine Alliance, a health partnership based in Geneva, Switzerland.

Note: this article says: “Studies have shown the P1 variant to be as much as 2.5 times more contagious than the original coronavirus ..”
That makes much more sense than the recent piece that claimed the B117 variant was 67 times more contagious than the original.
• Brazil’s P1 Coronavirus Variant Mutating, May Become More Dangerous (R.)
Brazil’s P1 coronavirus variant, behind a deadly COVID-19 surge in the Latin American country that has raised international alarm, is mutating in ways that could make it better able to evade antibodies, according to scientists studying the virus. Research conducted by the public health institute Fiocruz into the variants circulating in Brazil found mutations in the spike region of the virus that is used to enter and infect cells. Those changes, the scientists said, could make the virus more resistant to vaccines – which target the spike protein – with potentially grave implications for the severity of the outbreak in Latin America’s most populous nation.
“We believe it’s another escape mechanism the virus is creating to evade the response of antibodies,” said Felipe Naveca, one of the authors of the study and part of Fiocruz in the Amazon city of Manaus, where the P1 variant is believed to have originated. Naveca said the changes appeared to be similar to the mutations seen in the even more aggressive South African variant, against which studies have shown some vaccines have substantially reduced efficacy. “This is particularly worrying because the virus is continuing to accelerate in its evolution,” he added. Studies have shown the P1 variant to be as much as 2.5 times more contagious than the original coronavirus and more resistant to antibodies.
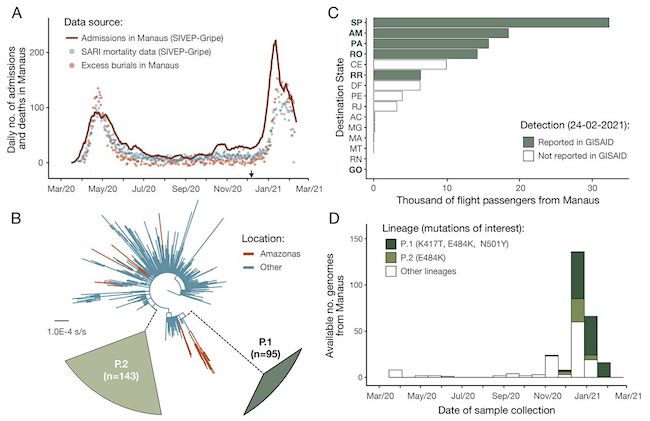

Can you still follow it?
• Norwegian Experts Recommend Stopping Further Use Of AstraZeneca Vaccine (RT)
The Institute of Public Health in Norway has recommended that the government stop using the Oxford/AstraZeneca Covid-19 vaccine after a lengthy review of the jab. Oslo suspended its use on March 11.
In a press release on Thursday, the agency said that after considering, in collaboration with external experts, the continued use of the heavily-scrutinized Covid-19 jab, that it would no longer recommend its use. It said more evidence has emerged to suggest there is a link between the jab and very rare cases of potentially fatal blood clots. “Based on this knowledge, we have arrived at a recommendation that the AstraZeneca vaccine be removed from the coronavirus vaccination program in Norway,” Geir Bukholm, director of infection control at the National Institute of Public Health, said in the statement.
The institute noted that Norway has made great strides in vaccinating its elderly and those most at risk of Covid. Therefore, those who are yet to be vaccinated are younger and less at risk from the virus – but are also potentially more prone to thrombotic events linked to the AstraZeneca vaccine. Those who have already received their first dose of the Anglo-Swedish jab will get a second shot from another vaccine manufacturer, the body stated. Also on Thursday, Health Minister Bent Hoeie said the government would not be drawn into a decision yet. “The government believes that we do not have a good enough basis for drawing a final conclusion that the AstraZeneca vaccine should be removed from the Norwegian vaccination program at this point,” Hoeie told a news conference.

They must have found a different science.
• Ireland May Delay 2nd Vaccine Doses Amid J&J Delays, AstraZeneca Age Limits (RT)
The Irish government has said it is considering whether to space out the administering of Pfizer’s Covid-19 vaccine in order to reach more people with a first dose as the country looks to open up from tough restrictions.
Speaking on RTÉ’s Morning Ireland on Thursday, the Irish deputy prime minister, Leo Varadkar, said it would make sense to space out the administering of the Pfizer shots, like in the UK, for those under 60 years old and in good health. The current interval between the administration of the two shots is 28 days, but some other countries have extended that to 12 weeks, allowing more people to be partially immunized against coronavirus in a shorter period of time. Research from the UK, before the country approved the Moderna jab, showed that the Pfizer and AstraZeneca vaccines greatly reduced the risk of hospitalization from Covid-19 after just one shot.
On Wednesday, Irish Health Minister Stephen Donnelly said a decision on whether to spread the vaccines out would be made within the week. The possible move comes as the EU’s medicines regulator reviews the use of the single-shot J&J vaccine and after Ireland limited the AstraZeneca vaccine to those aged 60 and over. On Tuesday, J&J said it would be delaying shipments to Europe while the vaccine is being reviewed following its suspension in the US; its rollout is yet to start in the EU. Similar technology was used in the development of their vaccines, which have been linked to very rare cases of blood clotting. Meanwhile, the EU announced on Wednesday that the bloc would be receiving an additional 25% of vaccines doses in quarter two from Pfizer.

It’s just lovely that every country’s experts reach different conclusions. Great for confidence.
• Poland Starts J&J Covid Vaccine Rollout Amid Objections, Health Concerns (RT)
Polish health authorities began rolling out doses of the Johnson & Johnson Covid-19 vaccine on Thursday, rejecting the concerns of the majority-Catholic country’s religious community and fears about the side effects of the jab. The government’s vaccine chief, Michal Dworczyk, confirmed the state’s vaccine rollout now included the Johnson & Johnson jab, stating health experts felt the benefits outweighed the side effects and they “have not received signals that there are problems or that patients are refusing [to take it].” Some 120,000 doses of the Johnson & Johnson vaccine have arrived in the country, with the first jabs from the consignment administered on Thursday.
On Wednesday, a senior official in Poland’s Catholic Church called on its members to reject both the Johnson & Johnson and AstraZeneca vaccines over the use of abortion-derived cells in their manufacturing processes. Bishop Jozef Wrobel declared that the church has a “serious moral objection” to the use of vaccines that include cells “derived from aborted foetuses” in the manufacturing process despite the jabs not containing any abortion-related cells in their ingredients. The comments from Poland’s religious authority contradict earlier remarks from Pope Francis, who declared that it is “morally acceptable” to be vaccinated with jabs developed using the aborted cell-linked manufacturing process. Addressing similar concerns to the Polish Catholic Church, the Pope said that having the vaccine does “not constitute formal cooperation” with or support of abortion.

“..if you have had a serious side effect and there is a temporal connection with the vaccination, then I would encourage you to seeks compensation from us..”
• 37 People In Denmark Seek Compensation Over Covid Vaccine Side Effects (RT)
Some 37 people in Denmark are requesting compensation over adverse effects they say were suffered after getting a Covid-19 jab. Most of the cases are related to AstraZeneca’s vaccine, which the country has stopped using. A total of 29 people have applied for compensation for side effects believed to be linked to the Swedish-British jab – which was renamed Vaxzevria last month – the Danish Patient Compensation Association said in a statement on Thursday. A further eight people applied for compensation in connection with the coronavirus jab made by Pfizer. The side effects cited in the applications range from mild fever and discomfort to such extreme conditions as paralysis, blood clots, miscarriage, and even death.
In Denmark, patients are eligible for state-sponsored compensations if they suffer rare or severe adverse effects of any medicines. The relatives of patients who have died can also receive compensation. “There are almost no drugs without side effects. That is why we have a safety net in Denmark that picks up the patients who are so unfortunate as to be affected by rare and serious side effects,” the director of Patient Compensation, Karen-Inger Bast, stated. The director also urged other people who might have suffered side effects to come forward with their complaints as well. She warned, though, that the health troubles must be serious in order to receive compensation.
“It is important to say that you do not receive compensation for mild and transient symptoms such as a little fever or pain in the arm. But if you have had a serious side effect and there is a temporal connection with the vaccination, then I would encourage you to seeks compensation from us,” Bast stated. The association’s announcement comes a day after Denmark became the first European country to completely discontinue use of AstraZeneca vaccine, citing “a possible link between very rare cases of unusual blood clots, bleeding, low blood platelets counts” and the shot by the drug maker. “Our overall assessment is there is a real risk of severe side effects associated with using the Covid-19 vaccine from AstraZeneca,” Danish Health Authority Director General Soeren Brostroem said.

Painting rapid tests in a bad light? PCR is crap too, so no testing left?
• Rapid Covid Testing In England May Be Scaled Back Over False Positives (G.)
Senior government officials have raised “urgent” concerns about the mass expansion of rapid coronavirus testing, estimating that as few as 2% to 10% of positive results may be accurate in places with low Covid rates, such as London. Boris Johnson last week urged everyone in England to take two rapid-turnaround tests a week in the biggest expansion of the multibillion-pound testing programme to date. However, leaked emails seen by the Guardian show that senior officials are now considering scaling back the widespread testing of people without symptoms, due to a growing number of false positives.
In one email, Ben Dyson, an executive director of strategy at the health department and one of health secretary Matt Hancock’s advisers, stressed the “fairly urgent need for decisions” on “the point at which we stop offering asymptomatic testing”. On 9 April, the day everyone in England was able to order twice-weekly lateral flow device (LFD) tests, Dyson wrote: “As of today, someone who gets a positive LFD result in (say) London has at best a 25% chance of it being a true positive, but if it is a self-reported test potentially as low as 10% (on an optimistic assumption about specificity) or as low as 2% (on a more pessimistic assumption).”
He added that the department’s executive committee, which includes Hancock and the NHS test and trace chief, Dido Harding, would soon need to decide whether requiring people to self-isolate before a confirmatory PCR test “ceases to be reasonable” in low infection areas where there is a high likelihood of a positive result being wrong. The accuracy of rapid coronavirus tests and how they should be deployed have been the focus of months of debate in the UK. The proportion of false positives – people incorrectly told they have the virus – increases when the prevalence of the disease falls. This happens because although the number of true positives is falling, the tests produce roughly the same number of false positives – meaning the proportion of incorrect results becomes greater.

Is it lockdowns, vaccines or 50% of under-25s already carrying antibodies because the virus was so widespread? Fried of a friend in London says everyone he knows has “already had it”.
• Is Boris Lauding Lockdowns Because He’s Planning Another For October? (Clark)
The prime minister’s claim that it was lockdown, and not the vaccine or seasonal factors, which led to the rapid fall in Covid cases and deaths in the UK should set alarm bells ringing very loudly. The Man in Black wanted us to be in no doubt during his interview this week with Sky News: it was the lockdown whatdunit. “It is very, very important for everybody to understand,” Johnson said, “that the reduction in these numbers, in hospitalisations, in deaths, in infections, has not been achieved because of the vaccination programme. “People don’t, I think, appreciate that it’s the lockdown that has been overwhelmingly important in delivering this improvement in the pandemic and in the figures that we’re seeing. And so, yes of course the vaccination programme has helped, but the bulk of the work in reducing the disease has been done by the lockdown.”
Repeat after Me: “The bulk of the work in reducing the disease has been done by the lockdown.. The bulk of the work…” Why is Johnson so keen to downplay the government’s roll-out of a mass vaccination programme and instead big-up lockdown? Some have said it’s because he fears legal challenges for locking us down again in early January and wants to get his defence statement in before the writ. But I think BoJo is looking forwards, not backwards. By stressing ’lockdowns work’ now, he is ‘nudging’ us towards accepting another shutdown of the non-virtual economy in autumn. There are already some pretty strong clues about what might lie ahead. As I’ve noted in previous articles, the extension of furlough has been an excellent guide over the last twelve months for what is going to happen – far better than the smug ‘inside the tent’ pundits who have assured us since last March that things would soon be getting back to normal.
In December, Chancellor Sunak’s extension of furlough until April was the giveaway – no pun intended – that we’d be locked down again in the New Year, and so it proved. On December 18, I correctly predicted the exact date (January 4) that Johnson would announce the new 2021 lockdown. In early March, furlough was extended again, until the end of September, despite the government assuring us all restrictions would be lifted on June 21. This suggests it’s odds-on that some form of restrictions will remain well past that date, because what happens in October? Why, the autumn/winter flu season starts. It would be so easy for Johnson at that point to say “We need another lockdown” to “protect the NHS” from another wave (the ‘third’ or ‘fourth’) or from a “variant of concern” of coronavirus.

They call it the “Digital Green Certificate” instead. Same difference.
• ‘Vaccine Passports’ Can’t Be Requirement For Travel, Says EU (F.)
Travel is on the up and up. CBS News Travel Editor, Peter Greenberg reported that U.S. domestic airfares are rapidly increasing, as are hotel rates (not to 2019 levels yet, but certainly rising). Cruise liners are not waiting for CDC permission to set sail from U.S. ports and are instead repositioning cruises to leave from and visit non-U.S. ports (in the Caribbean, for instance). One cruise liner started selling tickets for its 2021/2022 world cruise, at $40,o00 a cabin, and the ship sold out in one day. And of course, there needs to be a way to verify that people are traveling safely without transmitting the virus to one place from another. In the EU, all 27 countries have agreed that coordination is necessary and that this will take the form of a Digital Green Certificate.
Crucially, however, the EU has agreed that people who don’t have this certificate shouldn’t be discriminated against–that’s why it isn’t called a passport or doesn’t have anything called vaccine near the title. Firstly, it’s to be called a Digital Green Certificate, able to be downloaded onto your phones. It’s not called a vaccination passport, because–the EU said Thursday–you don’t need to be vaccinated to get it. You can also get a certificate if you have had Covid-19 (and recovered) and if you have had a negative Covid-19 test before departure. That way, it doesn’t discriminate between those that have been vaccinated and those who haven’t. As reported by The Local, the EU plans to roll it out by mid-June, initially across the 27 EU bloc to citizens and residents, but also eventually to be compatible with non-EU countries.
Eduardo Santander, chief executive officer of the European Travel Commission, an association of national tourism organizations based in Brussels said: “Finally, we have a tangible solution to coordinating and harmonizing travel measures. I think other countries like the U.S. will also come up with their own technological solutions that will be compatible, and after a period of trials this summer, a global standard will be established.” Individual countries will also have a way of setting individual rules within the certificate. For example, Hungary uses Russia’s Sputnik vaccine, which most of the other EU countries do not, and so–under the certificate–they would be able to incorporate the vaccines they each want to use.

We try to run the Automatic Earth on donations. Since ad revenue has collapsed, you are now not just a reader, but an integral part of the process that builds this site. Thank you for your support.



Support the Automatic Earth in virustime. Click at the top of the sidebars to donate with Paypal and Patreon.